- [email protected]
- 27-31 Wright St, Clayton VIC 3168
- Contact
Blog Details
-
EZADEH > My research > Cell-Based Genetic Diagnostic and Research Applications, In Situ Hybridisation for RNA and DNA detection
02May
Cell-Based Genetic Diagnostic and Research Applications, In Situ Hybridisation for RNA and DNA detection
by Elazadeh, 0 Comments

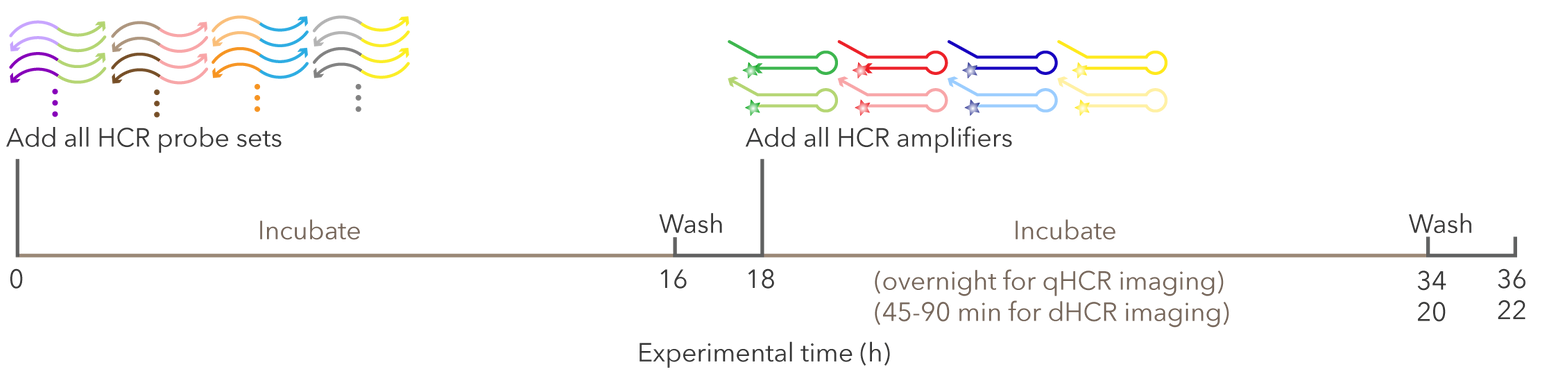
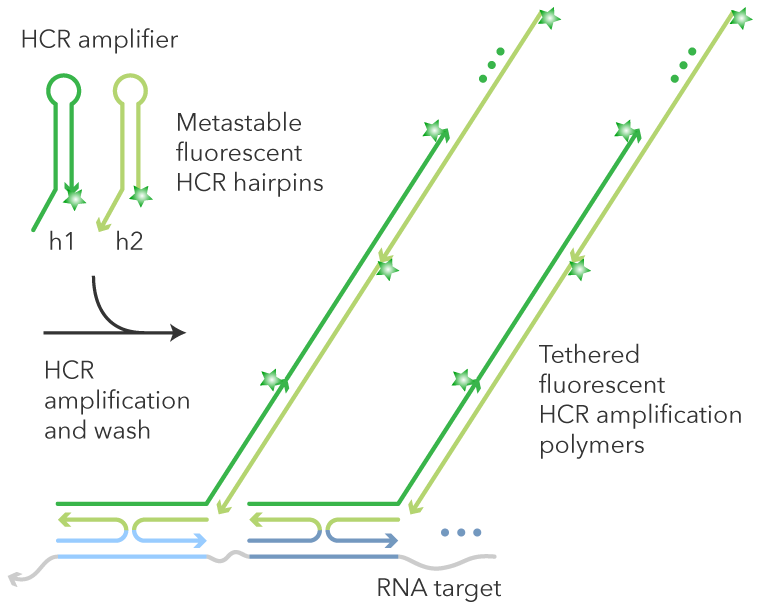
In Situ Hybridization (ISH)
In situ hybridisation (ISH) was first described in 1969 for localising specific DNA sequences on chromosomes in cytological preparations of Xenopus oocytes. Modifications of the original technique have been described that permit the detection and localisation of nucleic acids within tissues from a variety of different organisms. The target molecules of ISH are nucleic acids. Nucleic acids are complex with proteins in the cell; when tissue is embedded in a complex matrix, the nucleic acids are cross-linked to that matrix. Thus major challenges of ISH are to make target nucleic acid available to the probe; and once proper hybrids are formed, to stabilise them without destroying the cell morphology. Antigen reactivity (hybridisation) is based on complementarity between the sequence of interest and the designed probe, rather than on antigen-antibody recognition. Detection of the annealed products was originally based on the use of radiolabeled probes, which were visualised by slide emulsion autoradiography. Currently, especially in the clinical setting, radioisotopes have been replaced by non-isotopic detection methods. In chromogen-based ISH (CISH), a biotin-labelled or digoxigenin-labelled probe is detected with a secondary antibody and chromogenic detection.
Here our team design your primers (F, R) for the first step of the ISH and provide you customize protocols.
-
Tags:
- Laboratory techniques
